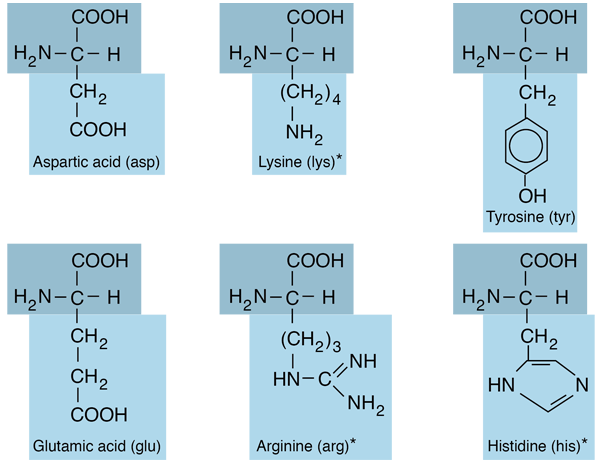

‘I kept on reading this and realising this wasn’t right,’ says Doig. There has been a tendency to see the choice of the 20 amino acids as arbitrary – as in the ‘frozen accident theory’ proposed by British molecular biologist Francis Crick in the 1960s, which suggested a different group of 20 would be just as good. The 20 amino acids have a range of properties…

The arrival of proteins and the eventual adoption of the standard 20 amino acids was likely to have been a big evolutionary step. By polymerising amino acids in long polypeptide chains, proteins could fold into soluble structures with close-packed cores and ordered binding pockets. He has a different take on the question: ‘ were chosen in the RNA world, where there had been life and metabolism for millions of years, already generating a vast number of organic molecules.’ If amino acids were a product of RNA metabolism this would hugely increase their concentrations in the environment, he argues.īut the selection of the 20 amino acids used in biology is clearly linked to the development of proteins. 2 Perhaps these generally simple and readily available amino acids were the first to be press-ganged into life?Īndrew Doig, a chemical biologist at the University of Manchester in the UK, has been thinking about the chemistry of evolution, when not carrying out his research into Alzheimer’s disease. 1 They are also found in meteorites: analysis of the Murichison meteorite, which landed in Australia in 1969, found at least 86 amino acids, substituent chains of up to nine carbon atoms and dicarboxyl and diamino functional groups. The famous Miller–Urey experiment from 1952 showed that with electric sparks simulating lightning, simple compounds like water, methane, ammonia and hydrogen would form well over 20 different amino acids. How the proteinogenic amino acids came to be on earth is another crucial question. But why amino acids? Bernd Moosmann, an expert in redox medicine at the Johannes Gutenberg University of Mainz in Germany suggests the first amino acids were used to anchor membranes to RNA structures: ‘You can see this even in modern life: DNA and RNA in bacteria and mitochondria are always attached from the inside to a membrane.’ Most researcher think this would have been occurring at least 4 billion years ago in an ‘RNA world’, where RNA molecules were the first self-replicators, as well as performing the catalytic role that proteins play today. They are the building blocks for proteins, biology’s workhorse macromolecules that provide structure and function in all organisms. Why 20 and not 10 or 30? And why those particular 20? Over the last few decades, the passionate chemists and molecular biologists who can’t leave these questions alone have started piecing together some convincing explanations.įrom alanine (A) to tyrosine (Y), 20 ‘proteinogenic’ amino acids, each abbreviated to a different initial, make up the alphabet soup of life. One fundamental question is why life is based on a set 20 amino acids. How chemistry could have brought us to complex life poses many open questions. But understanding evolution at the chemical level is their passion, even when funding is sparse. For many researchers, studying the chemical origins of life is a side project – it’s what they do in between their grant-funded work on the causes and cure of human disease.
